What is Airborne Surface Disinfection ?
ASD: safe and responsible disinfection by Devea
What is Airborne Surface Disinfection (ASD)?
Surface Disinfection can be either manual or airborne. Airborne Surface Disinfection (ASD) techniques bring three key advantages:
- They are reproducible;
- ASD doesn’t require a human presence;
- These techniques can uniformly reach all the surfaces of the volume to be treated, including inaccessible areas.
The disinfectant or biocide – hydrogen peroxide (H2O2) – is sprayed as a non-wetting micro-drop mist. Once the area to be treated is saturated, it settles on the surfaces and instantly kills bacteria, spores, viruses and yeasts. Hydrogen peroxide converts completely into H2O and O2 – our biocides are therefore 100% biodegradable. As there is no residue, the biocide has no residual effect. Airborne Surface Disinfection (ASD) therefore ponctually decontaminates an area without any residue.

ASD: A clear regulatory framework
The regulation requires efficacy tests to validate two important criteria: fog homogeneous diffusion and efficacy in biocharge reduction.
The efficacy of a diffusion can be evaluated in a simple and relatively fast way. Chemical and/or biological indicators are used to guarantee the efficiency of the biodecontamination and the safety of all users. The disinfection protocols designed with Devea are approved by the customers according to their specifications: reduction of microbial populations by 4-log10 (99.99%), 5-log10 (99.999%) or 6-log10 (99.9999%).
The ASD device/product pair must be approved (requirements of the EN 17272 standards of 2020). Disinfectants must not only comply with the BPR (Biocidal Products Regulation) Reg (EU) 528/2012, but also be registered and authorized for marketing. Devea is the only French ASD company to be granted a Marketing Authorization for its Phileas® solutions (MA FR-2019-0071). This Marketing Authorization validates the diffuser / biocide couple and the achieved results.
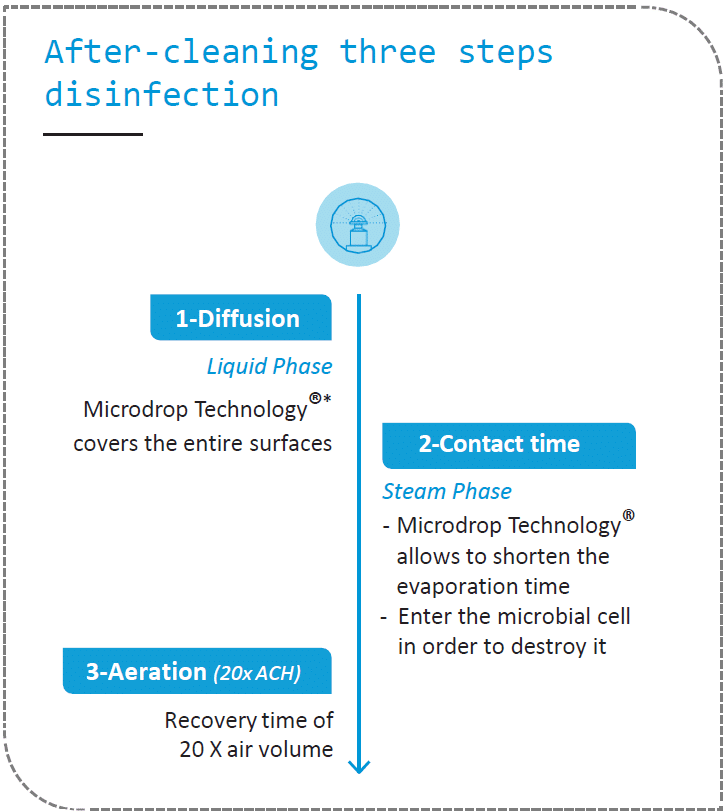
The key steps of the biodecontamination process
The validation of the ASD guarantees the restoration of the treated areas to a qualified state. This method of disinfection is part of a complete hygiene process called decontamination* with two main steps: cleaning* then disinfection* (see lexicon below). The cleaning phase is crucial as it removes organic and particulate residues and other chemical contaminants. Only a cleaned surface can be disinfected!